![HM Logo[words].jpg](https://static.wixstatic.com/media/f6e04a_f429f6c8934d4e4c9e79fcf44a95a0ff~mv2.jpg/v1/crop/x_15,y_114,w_628,h_360/fill/w_118,h_60,al_c,q_80,usm_0.66_1.00_0.01,enc_avif,quality_auto/HM%20Logo%5Bwords%5D.jpg)
DEFINING A NEW CATEGORY IN PREVENTIVE BONE HEALTH
Naquinate
Fracture prevention,
found in nature


The world's first biomarker-validated bone preservation supplement for the Osteopenic Window.
Working with the body to rebuild bone.
Haoma Medica is a science-driven biotech company with a pharmaceutical background, expanding into nutraceuticals through its subsidiary, Naquinate.
Naquinate is a naturally occurring compound that regulates both bone build-up and bone breakdown without side effects.

500M+
OSTEOPOROSIS
Diagnosed disease - pharmaceutical lane
1.8B+
OSTEOPENIC WINDOW
Sub-threshold - nutriceutical lane
15B+
BONE & JOINT SUPPLEMENT MARKET
Grow at a CAGR of 8.5% through 2035
THE OSTEOPENIC WINDOW
Affecting 40% of adults,
a diagnostic gap between healthy and diseased, clinically real and almost no one is treated.

ABOUT HAOMA MEDICA
A purpose-built biotech targeting the world's most under-treated bone disease.
Who We Are
Haoma Medica is a science-driven biotech company with a pharmaceutical background, expanding into nutraceuticals through its subsidiary, Naquinate. Founded by Prof. Stephen Hodges, Dr. Cenk Oguz and Carmen Greco, our team combines world-class academic science with deep clinical and commercial expertise.
Our Mission
To redefine bone health by delivering the world's first biomarker-validated supplement built on pharmaceutical-grade science — preventing fragility fractures before they happen.
Our Ingredient
Naquinate (Nq8) is a novel, small molecule that maintains the healthy balance between bone-building osteoblasts and bone-resorbing osteoclasts.
Academic Foundation
Our scientific programme is grounded in longstanding collaboration with The Royal Veterinary College, London, Tromsø University Hospital, and a network of internationally recognised advisors in bone metabolism, endocrinology, and clinical pharmacology.

THE PROBLEM
Osteoporosis: a global crisis hiding in plain sight.
37 million fragility fractures occur annually in individuals aged over 55, the equivalent of 70 fractures per minute. Yet more than 80% of at-risk individuals remain undiagnosed and untreated. Existing therapies carry significant side effects and compliance challenges.
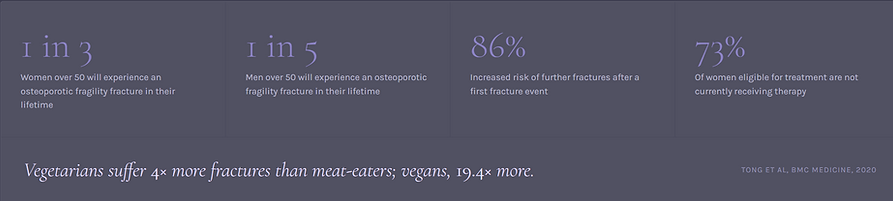

Why existing drugs fail patients.
Every approved osteoporosis therapy treats one side of bone biology — either resorption or formation. They are expensive, carry significant side effects, and are abandoned by most eligible patients. Anti-resorptive agents deliver only ~20% anti-fracture efficacy with little or no cortical effect.

INTRODUCING NAQUINATE Nq8
Developed at the Royal Veterinary College, London.

NaQuinate was developed by Prof. Stephen Hodges during collaborative research at the Royal Veterinary College. The compound is a synthetic derivative of vitamin K2 (menaquinone-7), engineered to maximise therapeutic bone activity while retaining the favourable safety profile of its parent compound.
The compound is covered by a robust intellectual property portfolio and has successfully progressed through chemical synthesis, in vitro biological activity characterisation, and two independent in vivo murine models of osteoporosis — demonstrating significant bone-protective effects in both models. Recent mechanistic work points to Wnt16-mediated β-catenin activation as a key driver of Naquinate's anabolic bone effect.
01 Phase I first-in-human trial completed — no significant safety or tolerability concerns
02 Demonstrated bone-protective efficacy in two independent murine osteoporosis models
03 Novel mechanism of action distinct from all currently approved osteoporosis therapies
04 Active collaboration with UCB, Royal Veterinary College, UCL and Tromsø University Hospital
Publications
70+ peer-reviewed publications from the Haoma Medica scientific team.
Patents
Granted patent covering UK, US, Canada, Australia, Japan, India, China, Brazil, Israel and the European zone.
Naquinate Nq8
Our supplement regulates both sides of bone remodelling.

A unique mechanism of dual action that occurs naturally within the body — Nq8 acts as a modulator of the constant regeneration cycle that maintains bone health, working directly on the two key bone cells.
01 Inhibits Osteoclasts
Suppresses the bone-resorbing cells that drive age-related bone loss — slowing the breakdown of existing bone tissue.
OPG/RANKL
Modulates osteoprotegerin (OPG) — the OPG/RANKL pathway is a recognised and established pathway in the bone remodelling process, controlling osteoclasts.
02 Promotes Osteoblasts
Simultaneously stimulates the cells that form new bone — restoring the formation/resorption balance disrupted in osteoporosis.
Wnt 16 / β-catenin
Modulates Wnt 16 — the Wnt 16/β-catenin pathway is a recognised and established pathway in the bone remodelling process, controlling osteoblasts.
Nq8 inhibits bone loss in a pharmaceutical industry “Gold Standard” animal model of postmenopausal osteoporosis.
The market is ready for a novel treatment with a new mechanism of action for managing patients at risk of fracture, one that addresses the unmet need for a therapy with greater efficacy in treating and preventing long-bone fractures, with a better safety profile. We believe Naquinate can meet these requirements.
01 Novel class of compound
02 Unique mechanism of action
03 A product naturally occurring in the body
04 Small molecule
05 Low dosage administered orally
Our products combine our novel bioactive ingredient Naquinate- Nq8 with a unique combination of ingredients that have been carefully formulated by our two founders, consultant orthopaedic surgeon Dr Cenk Oguz and bone health scientist Dr Stephen Hodges.

THE SCIENCE
Phase I Human Clinical Trial confirms safety & tolerability and Preclinical data confirms dual mechanism in vivo.
Both bone formation and bone strength increased in osteopenia and osteoporosis prevention models. The long limb bone (tibia) showed both expansion and positive change in thickness — and cortical effects were confirmed along the whole length of bone in repeat studies.
01
02
03
04
Cortical bone strengthened — long-term treatment increased cortical thickness and bone mineral density in the long limb bone.
Trabecular bone protected — short-term treatment (6 weeks) at 250 µg/Kg seen to protect trabecular bone in the proximal tibia.
Repeat study confirmed — cortical effects validated along the whole length of the long limb bone.
Dual modality observed — formation increased and resorption suppressed, consistent with mechanism.

FORMULATION
Naquinate, Nq8
Better bone health, scientifically formulated.
A new and unique combination of bone health supplements built around the novel dual bone action ingredient Nq8 — designed to strengthen bones, support normal density, minimise bone loss, prevent calcium build-up in arteries, and defend against fractures.
01 Complementary Ingredients
Carefully formulated synergy of cofactors that amplify Nq8 mechanism.
02 GMP Manufacture
Pharmaceutical-grade manufacturing under Good Manufacturing Practice.
03 Clean Labelling
No fillers, no animal by-products. Vegan, Kosher, Halal certified.
04 Sustainable Products
Plastic-free packaging and ingredient transparency throughout the supply chain.
05 Patent Protection
Supported by Haoma Medica's global patent portfolio covering composition and use

Further Ranges
Our future product pipeline targets different consumer markets, covering joint & cardiovascular health, sports nutrition, pet health, and a range of cosmeceutical products to maximise Nq8's wider product potential through additional mechanisms of action.
Further Opportunities
Emerging evidence suggests Nq8 reduces arterial calcification and inflammatory plaque, lowers IL-8 in liver cells to protect against fatty liver disease, may relieve gut inflammation in IBD, Crohn's and ulcerative colitis, and counters TGF-β to support kidney cell survival in age-related disease.
MANUFACTURING
Pharmaceutical rigour applied to a nutraceutical supply chain.

INVESTMENT ROUND
Investment round open Naquinate Nq8.

Haoma Medica is raising an Investment round to launch the Naquinate consumer brand and the physician-led Bone Preservation Assessment protocol.
US Market
Own the regulatory pathway to US market entry- Prepare and file the FDA NDI dossier to establish market readiness and regulatory credibility.
Physician Protocol
Activate practitioner advocacy as a trust engine- Build a US GP and physician channel to drive recommendations and clinical adoption. Include Bone Preservation Assessment protocol.
Science
Prove the science through expanded clinical validation- Advance Phase II trials to validate efficacy across cardiovascular, liver, renal, GI inflammatory pathology, skin ageing, and collagen health.
Regulatory
Prepare dossier and regulatory filings for the UK, Europe and RoW. Include an expansion and distribution blueprint.
Product
Build the commercial architecture for scale- Shape product strategy, manufacturing, packaging, pricing, and launch execution around premium growth.
Commercial
Engineer a multi-channel growth and licensing flywheel- Drive expansion through B2B licensing, distribution, geography, product innovation, influencer campaigns, and media demand.
